Question: The correct option with respect to the Pauling electronegativity values of the elements is :-
$\mathrm{Ga}<\mathrm{Ge}$
$\mathrm{Si}<\mathrm{Al}$
$\mathrm{P}>\mathrm{S}$
$\mathrm{Te}>\mathrm{Se}$
Correct Option: 1
Solution:
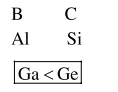
Along the period electronegativity increases