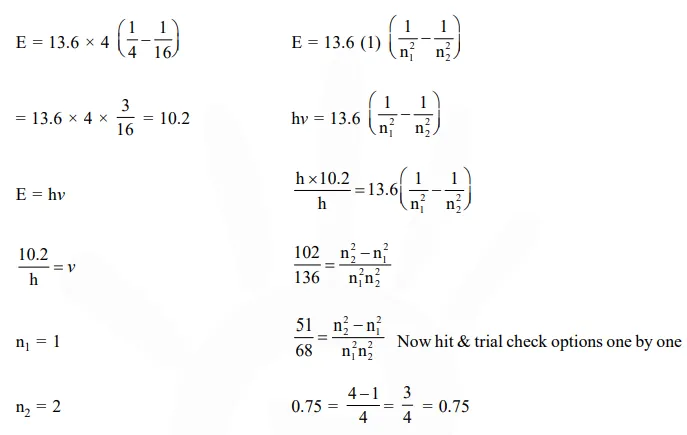The frequency of light emitted for the transition
Question:
The frequency of light emitted for the transition $\mathrm{n}=4$ to $\mathrm{n}=2$ of $\mathrm{He}^{+}$is equal to the transition in $\mathrm{H}$ atom corresponding to which of the following
Correct Option: , 2
Solution: