The P-V diagram of a diatomic ideal gas system going
Question:
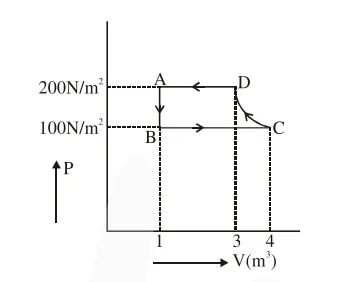
The P-V diagram of a diatomic ideal gas system going under cyclic process as shown in figure. The work done during an adiabatic process CD is (use $\gamma=1.4$ ) :
Correct Option: 1
Solution:
Adiabatic process is from $C$ to $D$
$\mathrm{WD}=\frac{\mathrm{P}_{2} \mathrm{~V}_{2}-\mathrm{P}_{1} \mathrm{~V}_{1}}{1-\gamma}$
$=\frac{200(3)-(100)(4)}{1-1.4}$
$=-500 \mathrm{~J}$
