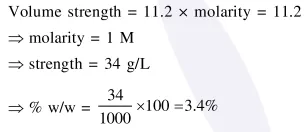The strength of
Question:
The strength of $11.2$ volume solution of $\mathrm{H}_{2} \mathrm{O}_{2}$ is : [Given that molar mass of $\mathrm{H}=1 \mathrm{~g} \mathrm{~mol}^{-1}$ and $\mathrm{O}=16 \mathrm{~g} \mathrm{~mol}^{-1}$ ]
Correct Option: , 2
Solution: