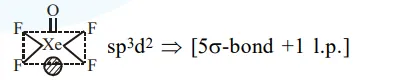The type of hybridisation and number of lone pair(s) of
Question:
The type of hybridisation and number of lone pair(s) of electrons of $\mathrm{Xe}$ in $\mathrm{XeOF}_{4}$, respectively, are :
Correct Option: , 3
Solution: