Three elements X, Y and Z are in the 3 period of the periodic table.
Question:
Three elements $X, Y$ and $Z$ are in the $3^{\text {rd }}$ period of the periodic table. The oxides of X, Y and Z, respectively, are basic, amphoteric and acidic. The correct order of the atomic numbers of $X, Y$ and $Z$ is :
Correct Option: , 2
Solution:
On moving left to right in a period, the acidic character of oxides increases.
$3^{\text {rd }}$ period element oxides.
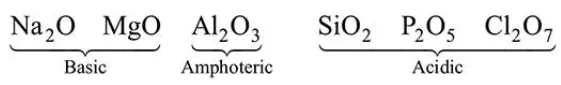
Acidic character $\propto$ Atomic No.
So, $\mathrm{X}$ have minimum atomic number while $\mathrm{Z}$ have maximum atomic number.
Thus, the correct order of the atomic number is
$X
