What does one mean by exothermic and endothermic reactions?
Question.
What does one mean by exothermic and endothermic reactions? Give examples.
What does one mean by exothermic and endothermic reactions? Give examples.
solution:
Chemical reactions that release energy in the form of heat, light, or sound are called exothermic reactions. Example: Reaction of sodium and chlorine to yield common salt
$\mathrm{Na}(\mathrm{s})+\frac{\mathbf{1}}{\mathbf{2}} \mathrm{Cl}_{2}(\mathrm{~s}) \rightarrow \mathrm{NaCl}(\mathrm{s})+411 \mathrm{~kJ}$ of energy
In other words, combination reactions are exothermic. Reactions that absorb energy or require energy in order to proceed are called endothermic reactions. For example: In the process of photosynthesis, plants use the energy from the sun to convert carbon dioxide and water to glucose and oxygen.
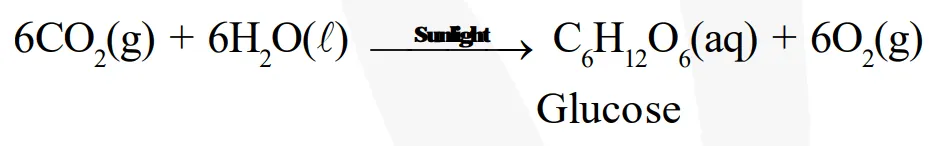
Chemical reactions that release energy in the form of heat, light, or sound are called exothermic reactions. Example: Reaction of sodium and chlorine to yield common salt
$\mathrm{Na}(\mathrm{s})+\frac{\mathbf{1}}{\mathbf{2}} \mathrm{Cl}_{2}(\mathrm{~s}) \rightarrow \mathrm{NaCl}(\mathrm{s})+411 \mathrm{~kJ}$ of energy
In other words, combination reactions are exothermic. Reactions that absorb energy or require energy in order to proceed are called endothermic reactions. For example: In the process of photosynthesis, plants use the energy from the sun to convert carbon dioxide and water to glucose and oxygen.

