Lyophilic and Lyophobic Colloids | Peptization for Class 12, IIT-JEE, NEET
Table of Contents
Colloidal solutions in which the dispersed phase has considerable affinity for the dispersion phase, are called Lyophilic sols (solvent-liking). For example - dispersion of gelatin starch, gum and proteins in water. Colloidal solutions in which the dispersed phase has no affinity or attraction for the medium or for the solvent are called Lyophobic colloidal (Solvent heating) solutions. Here, Get to know about Lyophilic and Lyophobic Colloids:
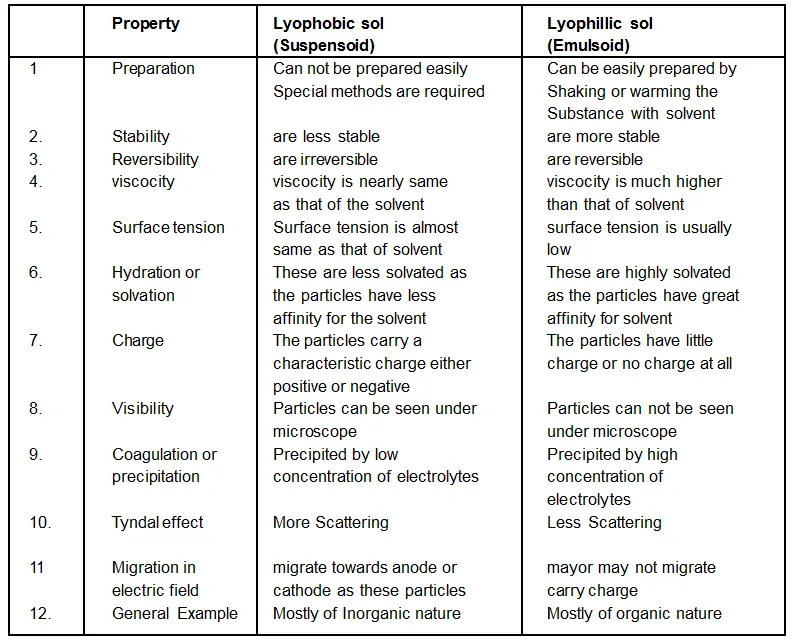
Difference between Lyophilic and Lyophobic Colloids
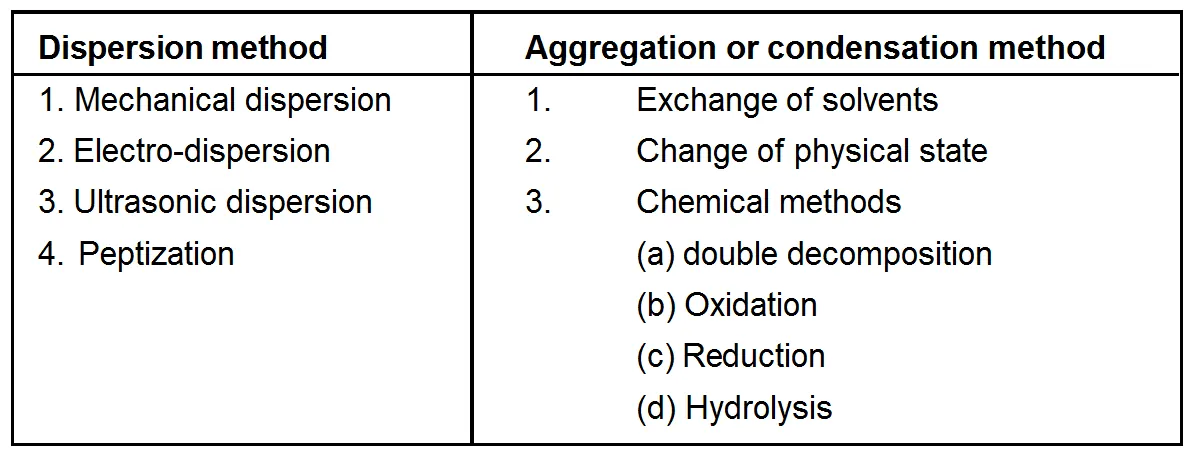
Method of Preparation
Bredig's Arc Method
Peptization Definition with Examples
 (b) Oxidation - A colloidal solution of Sulphur is obtained by passing into a solution of Sulphur dioxide.
(b) Oxidation - A colloidal solution of Sulphur is obtained by passing into a solution of Sulphur dioxide.
 Sulphur sol can also be obtained when is bubbled through an oxidizing agent (bromine water or Nitric acid)
(c) Reduction - Colloidal solutions of metals like gold, silver, platinum, lead etc. can be obtained when their salts solutions are act upon by reducing agents.
Sulphur sol can also be obtained when is bubbled through an oxidizing agent (bromine water or Nitric acid)
(c) Reduction - Colloidal solutions of metals like gold, silver, platinum, lead etc. can be obtained when their salts solutions are act upon by reducing agents.
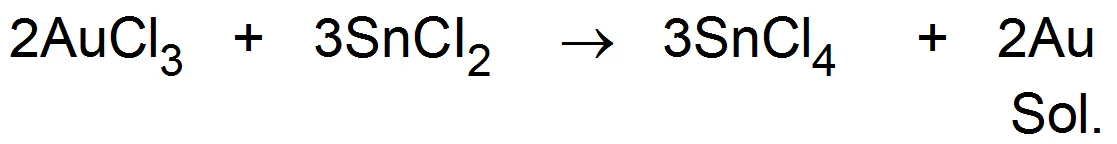
Difference between Lyophilic and Lyophobic Colloids

Methods of Preparation
Lyophilic sols may be prepared by simply warming the solid with liquid dispersion medium e.g. starch with water. On the other hand, Lyophobic sols have to be prepared by special methods. These methods fall into two categories (a) Dispersion Methods is which larger macro sized particles are broken down to colloidal size. (b) Condensation methods in which colloidal sized particles are built up by aggregating single ions or molecules. This method is also known as condensation method.
Electro-Dispersion (Bredig’s arc method)
This method is suitable for the preparation of colloidal solutions of metals like gold, silver, platinum etc, An arc is struck between the metal electrodes under, platinum etc, An arc is struck between the stabilizing agent such as a trace of KOH. The water is cooled by immersing the container in a ice bath. The intense heat of the arc vaporizes some of the metal which condenses under cold water. Note: 1. This method is not suitable when the dispersion medium is an organic liquid as considerable charring occurs. 2. This method comprises both dispersion and condensation.Peptization Definition
The dispersion of a freshly precipitated material into colloidal solution by the action of an electrolyte in solution is termed Peptization. The electrolyte used is called a Peptizing agent.A few examples of sols obtained by Peptization are:
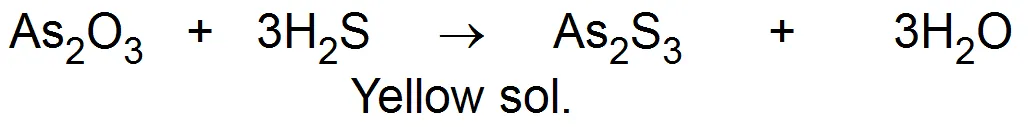
(i) Freshly prepared ferric hydroxide on treatment with a small amount of ferric chloride solution once forms a dark reddish-brown solution. Ferric chloride acts as a Peptizing agent. (ii) Freshly prepared stannic oxide on treatment with a small amount of dilute Hydrochloric acid forms a stable colloidal solution of stannic oxide. (iii) Freshly precipitated silver chloride can be converted into a colloidal solution by adding a small amount of Hydrochloric acid. (iv) Cadmium sulphide can be peptized with the help of hydrogen sulphide. The process of peptization thus involves the adsorption of suitable ions (supplied by the electrolyte added particularly a common ion) and electrically charged particles than split from the precipitate as colloidal particles. Chemical method: The chemical method involves chemical reactions in a medium in which the dispersed phase. is sparingly soluble. A condition of super-saturation is produced but the actual precipitation is avoided. Some familiar reactions used are: (a) Double decomposition (i) Arsenious sulphide sol: A 1 solution of arsenious oxide is prepared in hot water. The solution is cooled, filtered and is then gradually added to water saturated with hydrogen sulphide. whilst a stream of is being passed through the solution This is continued till an intense yellow colored solution is obtained. Excess of is removed by bubbling hydrogen through the solution.Click Here to Continue. . .
