A silver article generally turns black
A silver article generally turns black when kept in the open for a few days. The article when rubbed with toothpaste again starts shining.
(a) Why do silver articles turn black when kept in the open for a few days ? Name the phenomenon involved.
(b) Name the black substance formed and give its chemical formula.
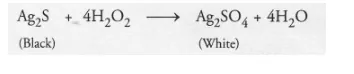
(a) Air contains traces of hydrogen sulphide (H2S) gas. It slowly reacts with silver present in the silver article to form silver sulphide (black in
colour). A coating of silver sulphide (Ag2S) gets deposited on the surface of the metal. The phenomenon is known as corrosion. Tooth pastes
generally contain traces of hydrogen peroxide as one of the constitutents. It reacts with silver sulphide to form silver sulphate which is white in
colour. Therefore, whiteness is restored to the article.
(b)