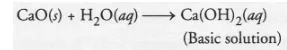Question:
A substance ‘A’, which is an oxide of a group 2 element, is used intensively in the cement industry. This element is present in bones also. On treatment
with water it forms a solution which turns red litmus blue. Identify A’ and also write the chemical reactions involved.
Solution:
The available information suggests that the substance A’ is oxide of the element calcium (Ca) which is present in group 2 of the periodic table.
Calcium is also a constituent of our bones in the form of calcium phosphate. . Calcium oxide (CaO) reacts with water to form calcium hydroxide
(basic in nature). It forms a basic solution which turns red litmus blue.