Apart from tetrahedral geometry, another possible geometry for CH4 is square planar
Apart from tetrahedral geometry, another possible geometry for CH4 is square planar with the four H atoms at the corners of the square and the C atom at its centre. Explain why CH4 is not square planar?
Electronic configuration of carbon atom:
${ }_{6} \mathrm{C} \cdot 1 s^{2} 2 s^{2} 2 p^{2}$
In the excited state, the orbital picture of carbon can be represented as:
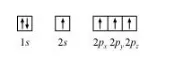
Hence, carbon atom undergoes sp3 hybridization in CH4 molecule and takes a tetrahedral shape.
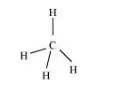
For a square planar shape, the hybridization of the central atom has to be dsp2. However, an atom of carbon does not have d-orbitalsto undergo dsp2 hybridization. Hence, the structure of CH4 cannot be square planar.
Moreover, with a bond angle of 90° in square planar, the stability of CH4 will be very less because of the repulsion existing between the bond pairs. Hence, VSEPR theory also supports a tetrahedral structure for CH4.
