Arrange the following metal complex/ compounds in the increasing order of spin only magnetic moment.
Arrange the following metal complex/ compounds in the increasing order of spin only magnetic moment. Presume all the three, high spin system.
(Atomic numbers $\mathrm{Ce}=58, \mathrm{Gd}=64$ and $\mathrm{Eu}=63 .$ )
(a) $\left(\mathrm{NH}_{4}\right)_{2}\left[\mathrm{Ce}\left(\mathrm{NO}_{3}\right)_{6}\right]$
(b) $\mathrm{Gd}\left(\mathrm{NO}_{3}\right)_{3}$ and
(c) $\mathrm{Eu}\left(\mathrm{NO}_{3}\right)_{3}$
Correct Option: , 4
(a) ${ }_{58} \mathrm{Ce} \rightarrow[\mathrm{Xe}] 4 \mathrm{f}^{2} 5 \mathrm{~d}^{0} 6 \mathrm{~s}^{2}$
In complex $\mathrm{Ce}^{4+} \rightarrow[\mathrm{Xe}] 4 \mathrm{f}^{0} 5 \mathrm{~d}^{0} 6 \mathrm{~s}^{0}$
there is no unpaired electron so $\mu_{\mathrm{m}}=0$
(b) ${ }_{64} \mathrm{Gd}^{3+} \rightarrow[\mathrm{Xe}] 4 \mathrm{f}^{7} 5 \mathrm{~d}^{0} 6 \mathrm{~s}^{0}$
contain seven unpaired electrons so,
$\mu_{\mathrm{m}}=\sqrt{7(7+2)}=\sqrt{63}$ B.M.
(c) ${ }_{63} \mathrm{Eu}^{3+} \rightarrow\left[{ }_{54} \mathrm{Xe}\right] 4 \mathrm{f}^{6} 5 \mathrm{~d}^{0} 6 \mathrm{~s}^{0}$
contain six unpaired electron
so, $\mu_{\mathrm{m}}=\sqrt{6(6+2)}=\sqrt{48}$ B.M.
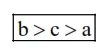
Hence, order of spin only magnetic movement