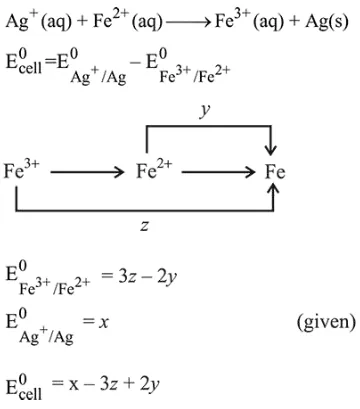
Question:
Calculate the standard cell potential (in V) of the cell in which following reaction takes place :
$\mathrm{Fe}^{2+}(\mathrm{aq})+\mathrm{Ag}^{+}(\mathrm{aq}) \rightarrow \mathrm{Fe}^{3+}(\mathrm{aq})+\mathrm{Ag}(\mathrm{s})$
Given that
$\mathrm{E}_{\mathrm{Ag}^{+} / \mathrm{Ag}}^{0}=x \mathrm{~V}$
$\mathrm{E}_{\mathrm{Fe}^{2+} / \mathrm{Fe}}^{0}=y \mathrm{~V}$
$\mathrm{E}_{\mathrm{Fe}^{3+} / \mathrm{Fe}}^{0}=z \mathrm{~V}$
Correct Option: , 3
Solution: