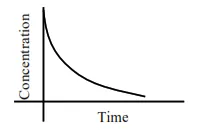
Question:
For the following graphs,
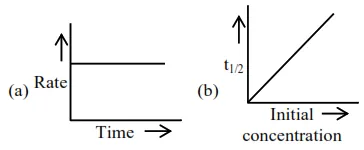
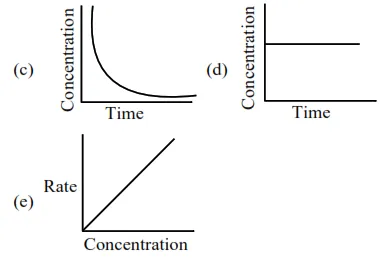
Choose from the options given below, the correct one regarding order of reaction is :
Correct Option: 1
Solution:
For zero order reaction's rate $=\mathrm{K}$ [Reactant] $^{0}$
$\Rightarrow \mathrm{r}=\mathrm{k}$
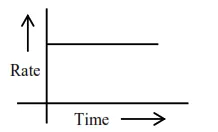
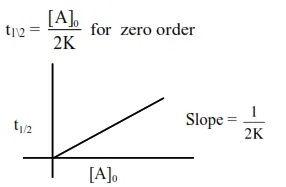
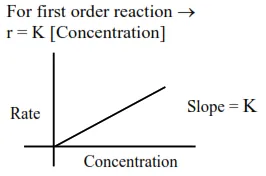
Reactant concentration after time $t \rightarrow$
$\mathrm{C}_{\mathrm{t}}=\mathrm{C}_{0} \mathrm{e}^{-\mathrm{kt}}$