Question:
Given below are two statements: one is labelled as Assertion A and the other is labelled as
Reason $\mathrm{R}$ :
Assertion $\mathbf{A}$ : The $\mathrm{H}-\mathrm{O}-\mathrm{H}$ bond angle in water molecule is $104.5^{\circ}$
Reason $\mathbf{R}$ : The lone pair - lone pair repulsion of electrons is higher than the bond pair - bond pair repulsion.
Correct Option: , 4
Solution:
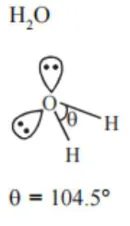
the hybridisation of oxygen is water molecule is $\mathrm{sp}^{3}$.
So electron geometry of water molecule is tetrahedral and the bond angle should be $109^{\circ} 28^{\prime \prime}$ but as we know that lone pair-lone pair repulsion of electrons is higher than the bond pair-bond pair repulsion because lone pair is occupied more space areound central atom than that of bond pair.
