In an attempt to demonstrate electrical conductivity
Question:
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus set up is given. Which among the following statement(s) is(are)
correct ?
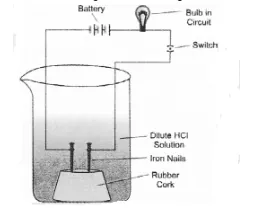
(i) Bulb will not glow because electrolyte is not acidic
(ii) Bulb will glow because HCl is a strong acid and furnishes ions for conduction.
(iii) Bulb will not glow because circuit is incomplete
(iv) Bulb will not glow because it depends upon the type of electrolytic solution
(a) (i) and (iii)
(b) (ii) and (iv)
(c) (ii) only
(d) (iv) only
Solution:
(c). Bulb will glow because the acid will furnish ions which conduct electricity.
