Question:
The increasing order of the boiling point of the major products $\mathrm{A}, \mathrm{B}$ and $\mathrm{C}$ of the following reactions will be:
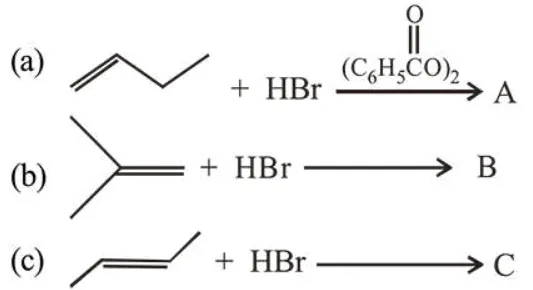
Correct Option: 1
Solution:
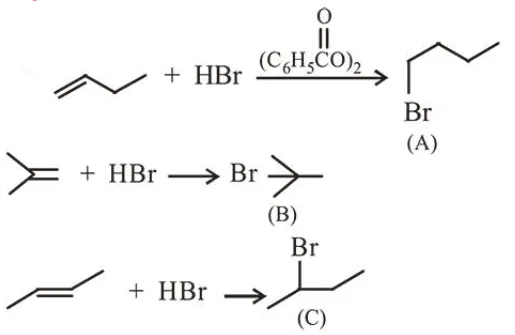
The boiling points of isomeric haloalkanes decrease with increase in branching.
So order of B.P. is A > C > B.
