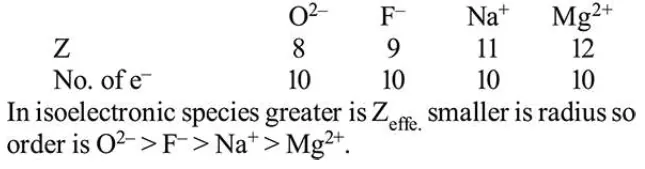Question: The ionic radii of $\mathrm{O}^{2-}, \mathrm{F}^{-}, \mathrm{Na}^{+}$and $\mathrm{Mg}^{2+}$ are in the order:
$\mathrm{F}^{-}>\mathrm{O}^{2-}>\mathrm{Na}^{+}>\mathrm{Mg}^{2+}$
$\mathrm{O}^{2-}>\mathrm{F}^{-}>\mathrm{Na}^{+}>\mathrm{Mg}^{2+}$
$\mathrm{Mg}^{2+}>\mathrm{Na}^{+}>\mathrm{F}^{-}>\mathrm{O}^{2-}$
$\mathrm{O}^{2-}>\mathrm{F}^{-}>\mathrm{Mg}^{2+}>\mathrm{Na}^{+}$
Correct Option: , 2
Solution: