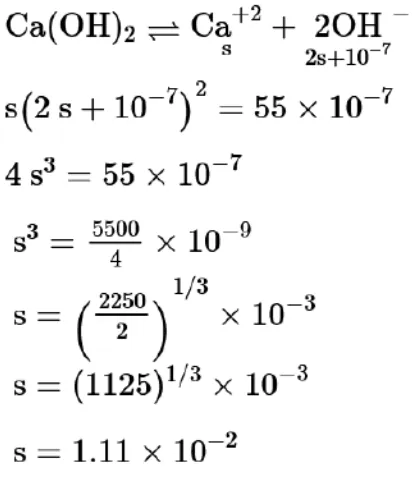Question: The solubility of $\mathrm{Ca}(\mathrm{OH})_{2}$ in water is :
[Given : The solubility product of $\mathrm{Ca}(\mathrm{OH})_{2}$ in water $=5.5 \times 10^{-6}$ ]
$1.11 \times 10^{-6}$
$1.77 \times 10^{-6}$
$1.77 \times 10^{-2}$
$1.11 \times 10^{-2}$
Correct Option: , 4
Solution: