Question:
What is the role of metal or reagents written on arrows in the given chemical reactions?
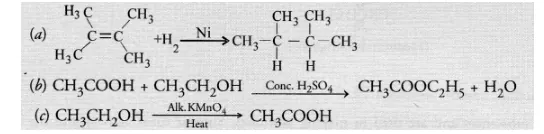
Solution:
(a) Nickel (Ni) acts as a hydrogenation catalyst for the reaction
(b) Cone. H2SO4 removes a molecule of H2O from the reaction mixture and acts as a dehydrating agent.
(c) Alkaline KMnO4 acts as an oxidising agent and oxidises ethanol to ethanoic acid.
