Salt Analysis - JEE Main Previous Year Questions with Solutions
JEE Main Previous Year Papers Questions of Chemistry with Solutions are available at eSaral. Practicing JEE Main chapter wise questions of Chemistry will help the JEE aspirants in realising the question pattern as well as help in analysing weak & strong areas.
Simulator
Previous Years AIEEE/JEE Mains Questions
Q. The products obtained on heating LiNO3 will be :-
(1) $\mathrm{LiNO}_{2}+\mathrm{O}_{2}$
(2) $\mathrm{Li}_{2} \mathrm{O}+\mathrm{NO}_{2}+\mathrm{O}_{2}$
(3) $\mathrm{Li}_{3} \mathrm{N}+\mathrm{O}_{2}$
(4) $\mathrm{Li}_{2} \mathrm{O}+\mathrm{NO}+\mathrm{O}_{2}$
AIEEE-2011
Ans. (2)
$\mathrm{LiNO}_{3} \longrightarrow \mathrm{Li}_{2} \mathrm{O}+\mathrm{NO}_{2}+\mathrm{O}$
Q. What is the best description of the change that occurs when $\mathrm{Na}_{2} \mathrm{O}(\mathrm{s})$ is dissolved in water ?
(1) Oxidation number of sodium decreases
(2) Oxide ion accepts sharing in a pair of electrons
(3) Oxide ion donates a pair of electrons
(4) Oxidation number of oxygen increases
AIEEE-2011
Ans. (3)
$\mathrm{Na}_{2} \mathrm{O}+\mathrm{HOH} \longrightarrow \mathrm{CaO}+\mathrm{CO}_{2}$
Q. Which of the following on thermal-decomposition yields a basic as well as an acidi oxide ?
(1) $\mathrm{NH}_{4} \mathrm{NO}_{3}$
(2) NaNO $_{3}$
(3) $\mathrm{KClO}_{3}$
(4) $\mathrm{CaCO}_{3}$
AIEEE-2012
Ans. (4)
$\mathrm{CaCO}_{3} \longrightarrow \mathrm{CaO}+\mathrm{CO}_{2}$
Q. Fire extinguishers contain H2SO4 and which one of the following :-
(1) $\mathrm{CaCO}_{3}$
(2) $\mathrm{NaHCO}_{3}$ and $\mathrm{Na}_{2} \mathrm{CO}_{3}$
(3) $\mathrm{Na}_{2} \mathrm{CO}_{3}$
(4) $\mathrm{NaHCO}_{3}$
JEE(Main)-2012 online-P-1
Ans. (4)
Fire extinguishers contain H2SO4 and NaHCO3
Q. Copper wire test for halogens is known as :-
(1) Duma's Test
(2) Beilstein's Test
(3) Lasssigne's Test
(4) Liebig's Test
JEE(Main)-2012 online-P-2
Ans. (2)
Beilstein's Test
Q. The standard potentials of $\mathrm{Ag}^{+} / \mathrm{Ag}, \mathrm{Hg}_{2}^{2+} / 2 \mathrm{Hg}, \mathrm{Cu}^{2+} / \mathrm{Cu}$ and $\mathrm{Mg}^{2+} /$ Mg electrodes are 0.80, 0.79, 0.34 and – 2.37 V, respectively. An aqueous solution which contains one mole per litre of the salts of each of the four metals is electrolyzed. With increasing voltage, the correct sequence of deposition of the metals at the cathode is :-
(1) Cu, Hg, Ag only
(2) Mg, Cu, Hg, Ag
(3) Ag, Hg, Cu only
(4) Ag, Hg, Cu, Mg
[JEE(Main)-2012 online-P-3]
Ans. (3)
Q. Beilstein test is used for estimation of which one of following elements ?
(1) S
(2) Cl
(3) C and H
(4) N
JEE(Main)-2012 online-P-3
Ans. (2)
$5 \mathrm{Mn} \mathrm{O}_{4}^{-}+2 \mathrm{C}_{2} \mathrm{O}_{4}^{2-}+16 \mathrm{H}^{+} \longrightarrow 5 \mathrm{Mn}^{+2}+2 \mathrm{CO}_{2}+\frac{16}{2} \mathrm{H}_{2} \mathrm{O}$
Q. In the following balanced reaction, $\mathrm{XMnO}_{+}^{-}+\mathrm{YC}_{2} \mathrm{O}_{4}^{2-}+\mathrm{ZH}^{+}=\mathrm{XMn}^{2+}+2 \mathrm{YCO}_{2}+$ $\frac{\mathrm{Z}}{2} \mathrm{H}_{2} \mathrm{O}$ the values of X, Y and Z, respectively are :-
JEE(Main)-2012 online-P-4
Ans. (2)
Q. A metal M on heating in nitrogen gas gives Y. Y on treatment with $\mathrm{H}_{2} \mathrm{O}$ gives a colourless gas which when passed through $\mathrm{CuSO}_{4}$ solution gives a blue colour, Y is :-
(1) $\mathrm{NH}_{3}$
(2) MgO
(3) $\mathrm{Mg}_{3} \mathrm{N}_{2}$
(4) $\mathrm{Mg}\left(\mathrm{NO}_{3}\right)_{2}$
JEE(Main)-2012 online-P-4
Ans. (3)
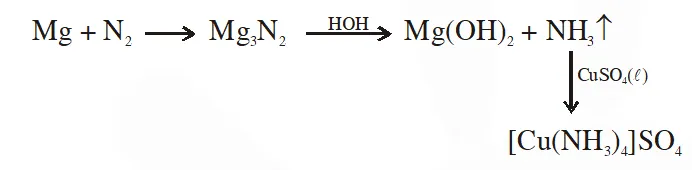
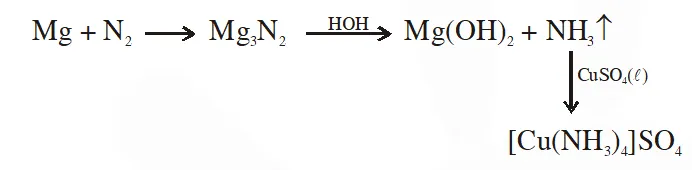
Q. Electrode potentials (E0) are given below :
$\mathrm{Cu}^{+} / \mathrm{Cu}=+0.52 \mathrm{V}, \mathrm{Fe}^{3+} / \mathrm{Fe}^{2+}=+0.77 \mathrm{V}$
$1 / 2 \mathrm{I}_{2}(\mathrm{s}) / \mathrm{I}^{-}=+0.54 \mathrm{V}, \mathrm{Ag}^{+} / \mathrm{Ag}=+0.88 \mathrm{V}$
Based on the above potentials, strnogest oxidizing agent will be :
(1) $\mathrm{Cu}^{+}$
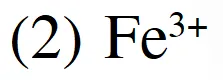 (3) $\mathrm{Ag}^{+}$
(4) $\mathrm{I}_{2}$
JEE(Main)-2013 online-P-1
(3) $\mathrm{Ag}^{+}$
(4) $\mathrm{I}_{2}$
JEE(Main)-2013 online-P-1
Ans. (3)
Q. Potassium dichromate when heated with concentrated sulphuric acid and a soluble chloride, gives brown - red vapours of:
( 1) $\mathrm{CrO}_{3}$
(2) $\mathrm{Cr}_{2} \mathrm{O}_{3}$
(3) $\mathrm{CrCl}_{3}$
(4) $\mathrm{CrO}_{2} \mathrm{Cl}_{2}$
JEE(Main)-2013 online-P-1
Ans. (4)
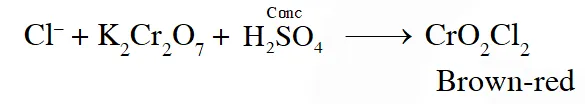
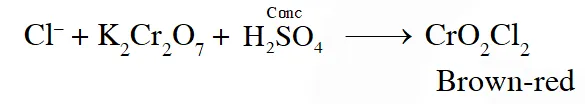
Q. Given :
$\overline{\mathrm{X}} \mathrm{Na}_{2} \mathrm{HAsO}_{3}+\mathrm{Y} \mathrm{NaBrO}_{3}+\mathrm{ZHCl} \rightarrow \mathrm{NaBr}+\mathrm{H}_{3} \mathrm{AsO}_{4}+\mathrm{NaCl}$
The values of X, Y and Z in the above redox reaction are respectively :
(1) 2, 1, 3 (2) 3, 1, 6 (3) 2, 1, 2 (4) 3, 1, 4
JEE(Main)-2013 online-P-1
Ans. (2)
$3 \mathrm{Na}_{2} \mathrm{HAsO}_{3}+\mathrm{NaBrO}_{3}+6 \mathrm{HCl} \longrightarrow \mathrm{NaBr}+\mathrm{H}_{3} \mathrm{AsO}_{4}+\mathrm{NaCl}$
Q. Sodium Carbonate cannot be used in place of $\left(\mathrm{NH}_{4}\right)_{2} \mathrm{CO}_{3}$ for the identification of $\mathrm{Ca}^{2+}, \mathrm{Ba}^{2+}$ and $\mathrm{Sr}^{2+}$ ions (in group V) during mixture analysis because :
(1) Sodium ions will react with acid radicals
(2) Concentration of $\mathrm{CO}_{3}^{2-}$ ions is very low
(3) Mg $^{2+}$ ions will also be precipitated
(4) $\mathrm{Na}^{+}$ ions will interfere with the detection of $\mathrm{Ca}^{2+}, \mathrm{Ba}^{2+}, \mathrm{Sr}^{2+}$ ions
JEE(Main)-2013 online-P-1
Ans. (3)
According to reactivity series Mg can be displaced by Na.
Q. Which of the following statements is incorrect?
(1) $\mathrm{Fe}^{2+}$ ion also gives blood red colour with $\mathrm{SCN}^{-}$ ion
(2) Cupric ion reacts with excess of ammonia solution to give deep blue colour of $\left[\mathrm{Cu}\left(\mathrm{NH}_{3}\right)_{4}\right]^{2+}$ ion.
(3) $\mathrm{Fe}^{3+}$ ion gives blood red colour with $\mathrm{SCN}^{-}$ ion.
(4) On passing $\mathrm{H}_{2} \mathrm{S}$ into $\mathrm{Na}_{2} \mathrm{ZnO}_{2}$ solution, a white ppt of ZnS is formed.
Ans. (1)
$\mathrm{Fe}^{+2}+\mathrm{SCN}^{-} \longrightarrow$ No colour
Q. Values of dissociation constant, $\mathrm{K}_{\mathrm{a}}$ are given as follows :
 (1) $\mathrm{NO}_{2}^{-}<\mathrm{CN}^{-}<\mathrm{F}^{-}$
(2) $\mathrm{F}^{-}<\mathrm{CN}^{-}<\mathrm{NO}_{2}^{-}$
(3) $\mathrm{NO}_{2}^{-}<\mathrm{F}^{-}<\mathrm{CN}^{-}$
(4) $\mathrm{F}^{-}<\mathrm{NO}_{2}^{-}<\mathrm{CN}^{-}$
JEE(Main)-2013 online-P-2
(1) $\mathrm{NO}_{2}^{-}<\mathrm{CN}^{-}<\mathrm{F}^{-}$
(2) $\mathrm{F}^{-}<\mathrm{CN}^{-}<\mathrm{NO}_{2}^{-}$
(3) $\mathrm{NO}_{2}^{-}<\mathrm{F}^{-}<\mathrm{CN}^{-}$
(4) $\mathrm{F}^{-}<\mathrm{NO}_{2}^{-}<\mathrm{CN}^{-}$
JEE(Main)-2013 online-P-2
 (1) $\mathrm{NO}_{2}^{-}<\mathrm{CN}^{-}<\mathrm{F}^{-}$
(2) $\mathrm{F}^{-}<\mathrm{CN}^{-}<\mathrm{NO}_{2}^{-}$
(3) $\mathrm{NO}_{2}^{-}<\mathrm{F}^{-}<\mathrm{CN}^{-}$
(4) $\mathrm{F}^{-}<\mathrm{NO}_{2}^{-}<\mathrm{CN}^{-}$
JEE(Main)-2013 online-P-2
(1) $\mathrm{NO}_{2}^{-}<\mathrm{CN}^{-}<\mathrm{F}^{-}$
(2) $\mathrm{F}^{-}<\mathrm{CN}^{-}<\mathrm{NO}_{2}^{-}$
(3) $\mathrm{NO}_{2}^{-}<\mathrm{F}^{-}<\mathrm{CN}^{-}$
(4) $\mathrm{F}^{-}<\mathrm{NO}_{2}^{-}<\mathrm{CN}^{-}$
JEE(Main)-2013 online-P-2
Ans. (4)
Order of basic strength according to Ka value.
$\mathrm{CN}^{-}>\mathrm{NO}_{2}^{-}>\mathrm{F}^{-}$
Q. Identify incorrect statement
(1) Copper (I) compounds are colourless except where colour results from charge transfer
(2) Copper (I) compounds are diamagnetic
(3) $\mathrm{Cu}_{2} \mathrm{S}$ is black
(4) $\mathrm{Cu}_{2} \mathrm{O}$ is colourless
JEE(Main)-2013 online-P-3
Ans. (4)
$\mathrm{Cu}_{2} \mathrm{O}$ is red.
Q. Given :
$\mathrm{E}_{1 / 2 \mathrm{C}_{2} / \mathrm{Cl}^{-}}^{0}=1.36 \mathrm{V}, \mathrm{E}_{\mathrm{Cr}^{3+} / \mathrm{Gr}}^{0}=-0.74 \mathrm{V} ; \quad \mathrm{E}_{\mathrm{G}_{2} \mathrm{O}_{7}^{2-} / \mathrm{Cr}^{3+}}^{0}=1.33 \mathrm{V}, \mathrm{E}_{\mathrm{MnO}_{4}^{-} / \mathrm{Mn}^{2+}}^{0}=1.51 \mathrm{V}$
The correct order of reducing power of the species $\left(\mathrm{Cr}, \mathrm{Cr}^{3+}, \mathrm{Mn}^{2+} \text { and } \mathrm{Cl}^{-}\right)$ will be :
JEE(Main)-2013 online-P-3
(1) $\mathrm{Mn}^{2+}<\mathrm{Cl}^{-}<\mathrm{Cr}^{3+}<\mathrm{Cr}$
(2) $\mathrm{Cr}^{3+}<\mathrm{Cl}^{-}<\mathrm{Mn}^{2+}<\mathrm{Cr}$
(3) $\mathrm{Cr}^{3+}<\mathrm{Cl}^{-}<\mathrm{Cr}<\mathrm{Mn}^{2+}$
(4) $\mathrm{Mn}^{2+}<\mathrm{Cr}^{3+}<\mathrm{Cl}^{-}<$
Ans. (1)
Order of reducing power acoording to E
$\mathrm{Cr}>\mathrm{Cr}^{+3}>\mathrm{Cl}^{-}>\mathrm{Mn}^{+2}$
Q. Which one of the following cannot function as an oxidising agent ?
(1) $\mathrm{NO}_{3}^{-}(\mathrm{aq})$
( 2) $\mathrm{I}^{-}$
(3) $\mathrm{Cr}_{2} \mathrm{O}_{7}^{2-}$
( 4) $\mathrm{S}_{(\mathrm{S})}$
JEE(Main)-2014 online_P-2
Ans. (2)
$\mathrm{I}^{-}$ has lowest oxidation state.
Q. Which of the following statements about $\mathrm{Na}_{2} \mathrm{O}_{2}$ is not correct ?
(1) $\mathrm{Na}_{2} \mathrm{O}_{2}$ oxidises $\mathrm{Cr}^{3+}$ to $\mathrm{CrO}_{4}^{2-}$ in acid medium
(2) It is diamagnetic in nature
(3) It is the super oxide of sodium
(4) It is a derivative of $\mathrm{H}_{2} \mathrm{O}_{2}$
JEE(Main)-2014 online_P-2
Ans. (2)
$\mathrm{Na}_{2} \mathrm{O}_{2}$ is peroxide
Q. Given :-
$\mathrm{Fe}^{3+}(\mathrm{aq})+\mathrm{e}^{-} \rightarrow \mathrm{Fe}^{2+}(\mathrm{aq}) ; \mathrm{E}^{\circ}=+0.77 \mathrm{V}$
$\mathrm{A}^{3+}(\mathrm{aq})+3 \mathrm{e}^{-} \rightarrow \mathrm{Al}(\mathrm{s}) ; \mathrm{E}^{\circ}=-1.66 \mathrm{V}$
$\mathrm{Br}_{2}(\mathrm{aq})+2 \mathrm{e}^{-} \rightarrow 2 \mathrm{Br}^{-} ; \mathrm{E}^{\circ}=+1.09 \mathrm{V}$
Considering the electrode potentials, which of the following represents the correct order of reducing power?
(1) $\mathrm{Al}<\mathrm{Fe}^{2+}<\mathrm{Br}^{-}$
(2) $\mathrm{Al}<\mathrm{Br}^{-}<\mathrm{Fe}^{2+}$
(3) $\mathrm{Fe}^{2+}<\mathrm{Al}<\mathrm{Br}^{-}$
$(4) \mathrm{Br}^{-}<\mathrm{Fe}^{2+}<\mathrm{Al}$
JEE(Main)-2014 online-P-2
Ans. (4)
Order of reducing power according to $\mathrm{E}^{\circ}$ $\mathrm{Br}^{-}<\mathrm{Fe}^{+2}<\mathrm{Al}$
Q. Consider the following equilibrium
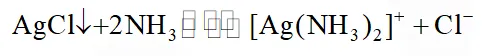 White precipitate of AgCl appears on adding which of the following?
(1) $\mathrm{NH}_{3}$
(2) Aqueous NaCl
(3) Aqueous $\mathrm{NH}_{4} \mathrm{Cl}$
(4) AqueousHNO $_{3}$
JEE(Main)-2014 online-P-2
White precipitate of AgCl appears on adding which of the following?
(1) $\mathrm{NH}_{3}$
(2) Aqueous NaCl
(3) Aqueous $\mathrm{NH}_{4} \mathrm{Cl}$
(4) AqueousHNO $_{3}$
JEE(Main)-2014 online-P-2
Ans. (4)
$\left[\mathrm{Ag}\left(\mathrm{NH}_{3}\right)_{4}\right] \mathrm{Cl}+\mathrm{aqH} \mathrm{NO}_{3} \longrightarrow \mathrm{AgCl} \downarrow$
Q. Copper becomes green when exposed to moist air for a long period. This is due to :-
(1) the formation of a layer of cupric oxide on the surface of copper.
(2) the formation of basic copper sulphate layer on the surface of the metal
(3) the formation of a layer of cupric hydroxide on the surface of copper.
(4) the formation of a layer of basic carbonate of copper on the surface of copper.
JEE(Main)-2014 online-P-3
Ans. (4)
$\mathrm{Cu} \frac{\text { Moist }}{\mathrm{air}}>\mathrm{CuCO}_{3} \cdot \mathrm{Cu}(\mathrm{OH})_{2}$
Q. The correct statement for the molecule, $\mathrm{CsI}_{3},$ is:
(1) it contains $\mathrm{Cs}^{3+}$ and $\mathrm{I}^{-}$ ions
(2) dit contains $\mathrm{Cs}^{3+}$ and $\mathrm{I}^{-}$ ions
(3) it is a covalent molecule
(4) it contains $\mathrm{Cs}^{+}$ and $\mathrm{I}_{3}^{-}$ ions
JEE(Main)-2014
Ans. (4)
$\mathrm{CsI}_{3} \longrightarrow \mathrm{Cs}^{+}+\mathrm{I}_{3}^{-}$
Q. Which one of the following exhibits the largest number of oxidation states ?
(1) Mn(25)
(2) V(23)
(3) Cr (24)
(4) Ti (22)
JEE(Main)-2014 online-P-3
Ans. (1)
Mn has maximum number of oxidation state.
Q. Hydrogen peroxide acts both as an oxidising and as a reducing agent depending upon the nature of the reacting species. In which of the following cases $\mathrm{H}_{2} \mathrm{O}_{2}$ acts as a reducing agent in acid medium ? :-
JEE(Main)-2014 online-P-3
(1) $\mathrm{MnO}_{4}^{-}$
(2) $\mathrm{SO}_{3}^{2-}$
(3) KI
(4) $\mathrm{Cr}_{2} \mathrm{O}_{7}^{2-}$
Ans. (1)
$\mathrm{H}_{2} \mathrm{O}_{2}$ acts as a reducing agent in acidic medium in pressure of $\mathrm{MnO}_{4}^{-}$
Q. Consider the reaction
$\mathrm{H}_{2} \mathrm{SO}_{3(\mathrm{aq})}+\mathrm{Sn}_{(\mathrm{aq})}^{4+}+\mathrm{H}_{2} \mathrm{O}_{(\mathrm{l})} \rightarrow \mathrm{Sn}_{(\mathrm{aq})}^{2+}+\mathrm{HSO}_{4(\mathrm{aq})}^{-}+3 \mathrm{H}_{(\mathrm{aq})}^{+}$
Which of the following statements is correct?
(1) $\mathrm{H}_{2} \mathrm{SO}_{3}$ is the reducing agent because it undergoes oxidation
(2) $\mathrm{H}_{2} \mathrm{SO}_{3}$ is the reducing agent because it undergoes reduction
(3) $\mathrm{Sn}^{4+}$ is the reducing agent because it undergoes oxidation
(4) $\mathrm{Sn}^{4+}$ is the oxidizing agent because it undergoes oxidation
JEE(Main)-2014 online-P-4
Ans. (1)
Q. How many electrons are involved in the following redox reaction ?
$\mathrm{Cr}_{2} \mathrm{O}_{7}^{2-}+\mathrm{Fe}^{2+}+\mathrm{C}_{2} \mathrm{O}_{4}^{2-} \rightarrow \mathrm{Cr}^{3+}+\mathrm{Fe}^{3+}+\mathrm{CO}_{2}$ (Unbalanced)
(1) 3 (2) 4 (3) 5 (4) 6
JEE(Main)-2014 online-P-4
Ans. (4)
$6 e^{-}$ are involved in this reaction
Q. Amongst the following, identify the species with an atom in +6 oxidation state:
(1) $\left[\mathrm{MnO}_{4}\right]^{-}$
(2) $\left[\mathrm{Cr}(\mathrm{CN})_{6}\right]^{3-}$
(3) $\mathrm{Cr}_{2} \mathrm{O}_{3}$
(4) $\mathrm{Cr} \mathrm{o}_{2} \mathrm{Cl}_{2}$
JEE(Main)-2014 online-P-4
Ans. (4)
$\mathrm{CrO}_{2} \mathrm{Cl}_{2} \mathrm{x}+(-4)+(-2)=0$
$\mathrm{x}=+6$
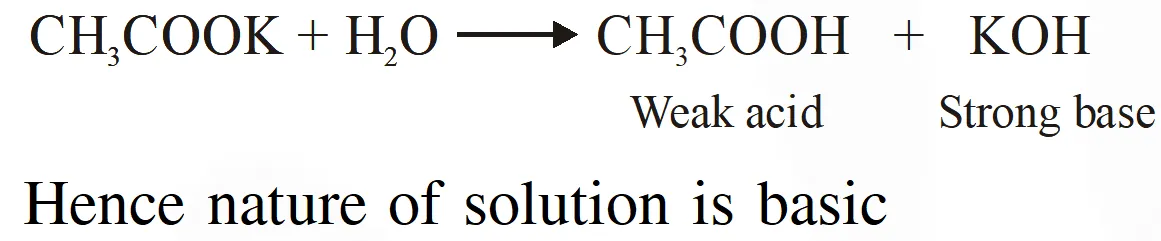
Q. Which of the following salts is the most basic in aqueous solution ?
( 1) $\mathrm{CH}_{3} \mathrm{COOK}$
( 2) $\mathrm{FeCl}_{3}$
(3) $\mathrm{Pb}\left(\mathrm{CH}_{3} \mathrm{COO}\right)_{2}$
(4) $\mathrm{Al}(\mathrm{CN})_{3}$
JEE(Main)-2018
Ans. (1)